38 mo diagram for b2
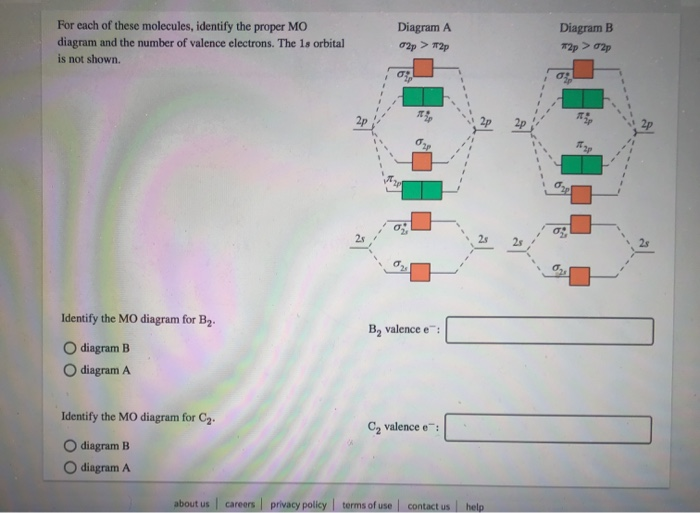
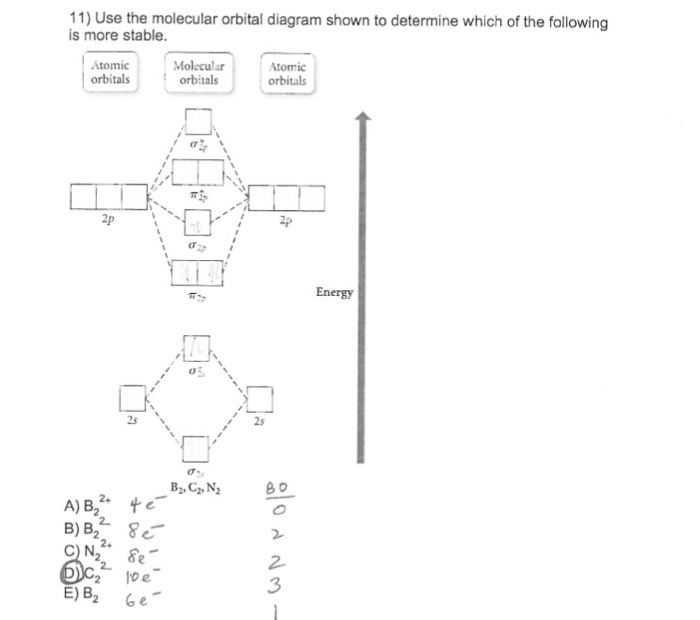
H2^- 4. H2^+ 5. B2^- Q. Use the MO diagram in Figure 7.16a in the textbook to describe the bonding in N2^+, N2 and N2^-. What is the bond order of each? Bond order for N2^+ Q. ... Our tutors rated the difficulty of Use the molecular orbital diagram shown to determine which o... as medium difficulty. The bond order of B2, C2, and N2 are 1, 2, and 3, respectively. B2 has two unpaired electrons with the same spin and therefore is paramagnetic. C2 and N2 are...
"BO" = 1/2 Boron atom is atomic number 5 in the periodic table, so it has five electrons. Thus, B_2 carries ten total electrons. The atomic orbitals each boron contributes consists of the 1s, 2s, and 2p. The ns orbitals combine to give a portion of the molecular orbital (MO) diagram like this: where sigma^"*" indicates an antibonding sigma (sigma) MO, and sigma is the bonding MO.
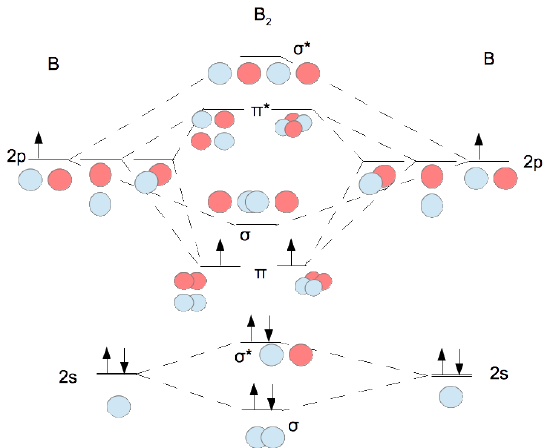
Mo diagram for b2
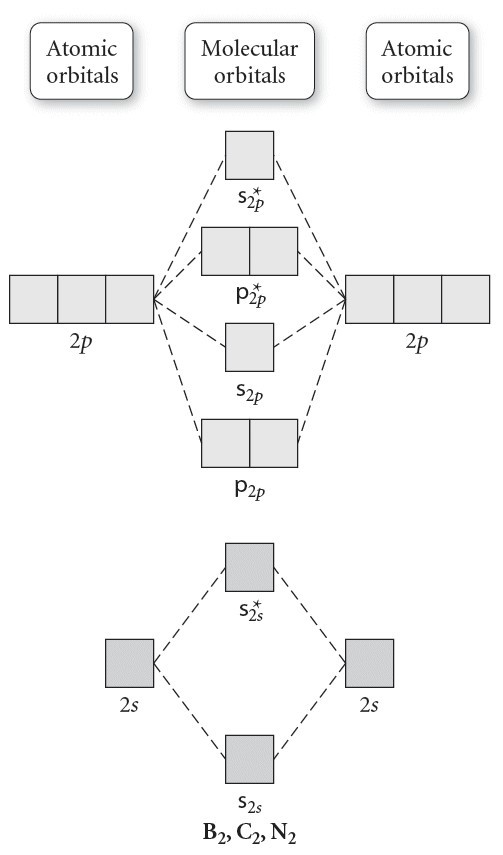
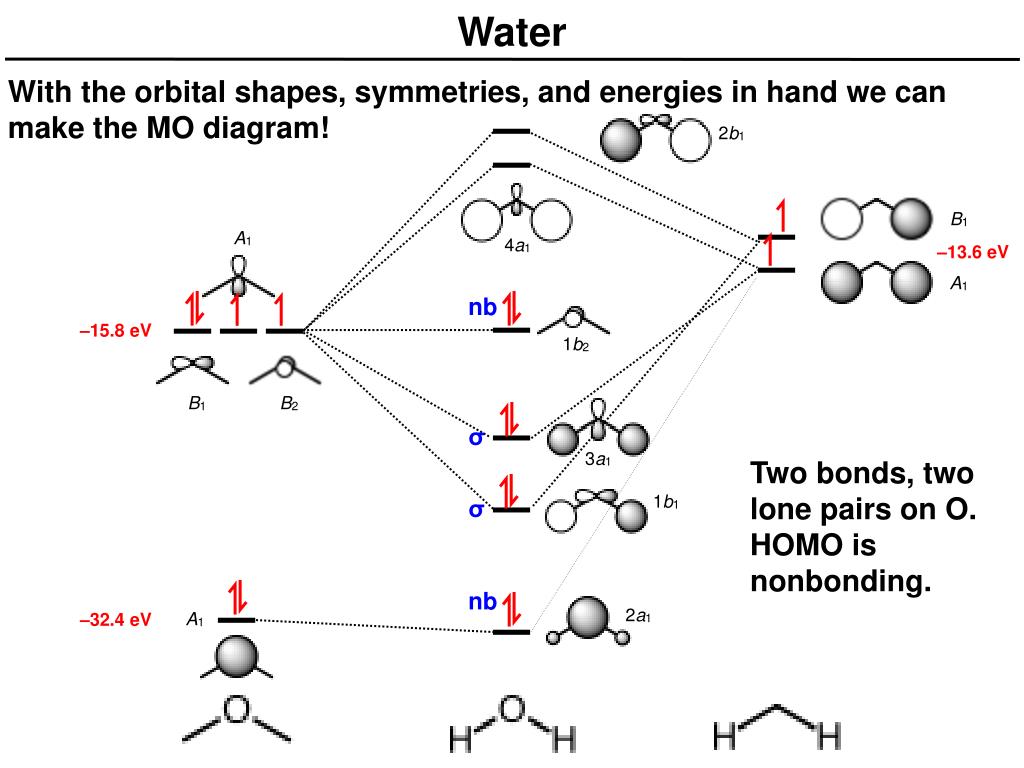
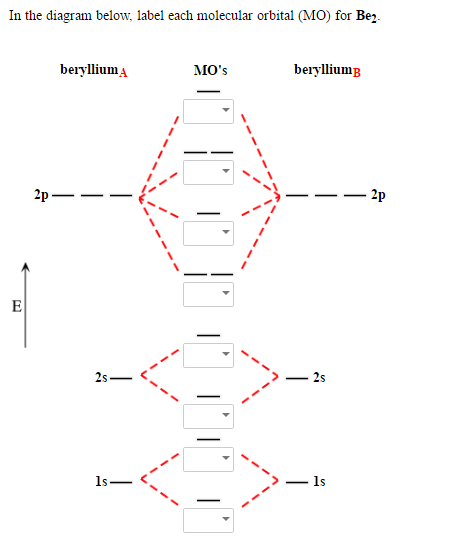
Molecular Orbital (MO) Theory helps us to explain and understand certain Part B – Molecular Orbital Energy Diagrams & Bond Order . + and Be2.A molecular orbital diagram, or MO diagram, is a qualitative descriptive tool explaining chemical bonding in molecules in terms of molecular orbital theory in general and the linear combination of atomic ... Relative AO Energies for MO Diagrams H He Li Be B C N O F Ne B C N O F Ne Na Mg Al Si P S Cl Ar Al Si P S Cl Ar 1s 2s 2p 3s 3p –19.4 eV –15.8 eV –32.4 eV –10.7 eV Photoelectron spectroscopy gives us a pretty good idea of the relative energies for AOs. Before we can draw a molecular orbital diagram for B₂, we must find the in-phase and out-of-phase overlap combinations for boron's atomic orbitals. Then we rank them in order of increasing energy. We can ignore the 1s orbitals, because they do not contain the valence electrons. Each boron atom has one 2s and three 2p valence orbitals. The 2s orbitals will overlap to form 2sσ and 2sσ ...
Mo diagram for b2. Re: M.O. Diagram for B2 Post by Chem_Mod » Tue Nov 11, 2014 11:21 pm As discussed in class the MO diagram for B 2 shows that it has two unpaired electrons (which makes it paramagnetic) and these electrons are in bonding molecular orbitals resulting in the equivalent bond strength of one bond. Draw the MO diagram for the B2 molecule and answer the following questions: A. Based on your MO diagram, would you expect the B2 molecule to be paramagnetic or diamagnetic? Explain your reasoning. B. Based on your MO diagram, do you think the B2-ion would have a stronger or weaker bond than the B2 molecule? Explain your reasoning. #3. Draw the MO diagram for `O_2^+` This is a bit of a curveball, but a perfectly valid problem. Recall that a cation indicates a loss of `1` electron. `O_2^+` is just the ionized form of `O_2`; that is, it's `O_2` with `1` missing electron. The MO diagram will be the same as the MO diagram of `O_2`, except with `1` less electron. MO electronic configuration: Bond order: Here Nb = 4, Na = 2 Bond order = The two boron atom is B2 molecules are linked by one covalent bond. Magnetic properties: Since each 2p x and 2p y MO contains unpaired electron, therefore B 2 molecule is paramagnetic.
Before we can draw a molecular orbital diagram for B₂, we must find the in-phase and out-of-phase overlap combinations for boron's atomic orbitals. Then we rank them in order of increasing energy. We can ignore the 1s orbitals, because they do not contain the valence electrons. Each boron atom has one 2s and three 2p valence orbitals. The 2s orbitals will overlap to form 2sσ and 2sσ ... Relative AO Energies for MO Diagrams H He Li Be B C N O F Ne B C N O F Ne Na Mg Al Si P S Cl Ar Al Si P S Cl Ar 1s 2s 2p 3s 3p –19.4 eV –15.8 eV –32.4 eV –10.7 eV Photoelectron spectroscopy gives us a pretty good idea of the relative energies for AOs. Molecular Orbital (MO) Theory helps us to explain and understand certain Part B – Molecular Orbital Energy Diagrams & Bond Order . + and Be2.A molecular orbital diagram, or MO diagram, is a qualitative descriptive tool explaining chemical bonding in molecules in terms of molecular orbital theory in general and the linear combination of atomic ...










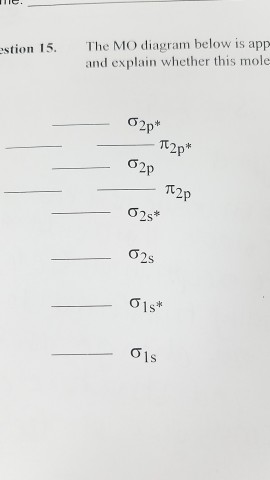










0 Response to "38 mo diagram for b2"
Post a Comment